Ceramic, Heal Thyself


 Behold the routine finger cut.
Behold the routine finger cut.
What happens next?
Your finger bleeds. There is slight swelling. Time passes, the swelling goes down, and the blood coagulates and generates new skin.
Presto: Your skin has healed itself.
“Look at this,” said Priya Vashishta, holding up a butter plate while dining on the patio at one of his favorite downtown L.A. spots, Café Pinot.
“If I drop this plate, it will shatter,” said Vashishta, a professor of materials science, computer science, and physics and astronomy at USC Viterbi and director of the Collaboratory for Advanced Computing and Simulations.
But can the ceramic plate put itself back together — heal itself — much like a cut finger?
Well, not exactly.
But when made with a special additive, and with a little help from heat, ceramic products can heal surface microcracks on their own — making the products more durable and long lasting.
The research Vashishta and his team are conducting on self-healing materials has military and industrial applications.
For example, the rotors of military helicopters now are safer because they include self-healing ceramic ball bearings that last longer and are more durable than steel ball bearings, Vashishta said.
After, say, 1,000 hours of use, the ball bearings are X-rayed and if microcracks are detected, the ball bearings can be heated to a degree that would melt steel to kick-start the process of self-repairing.
Ceramic can withstand twice the amount of heat as steel.
For self-healing to occur, ceramic products must include nanoparticles of silicon carbide (a ceramic material nearly as hard as diamond) that, when heated and oxygenated, can mimic nature.
“This happens roughly in the same way a cut finger heals itself,” Vashishta said.
For healing to occur on a finger, in steel, or ceramics, some material needs to move, fill a gap, and solidify or coagulate, Vashishta explained. Self-healing is easiest in skin and other soft materials, more difficult in steel and even more difficult in ceramics, he said.
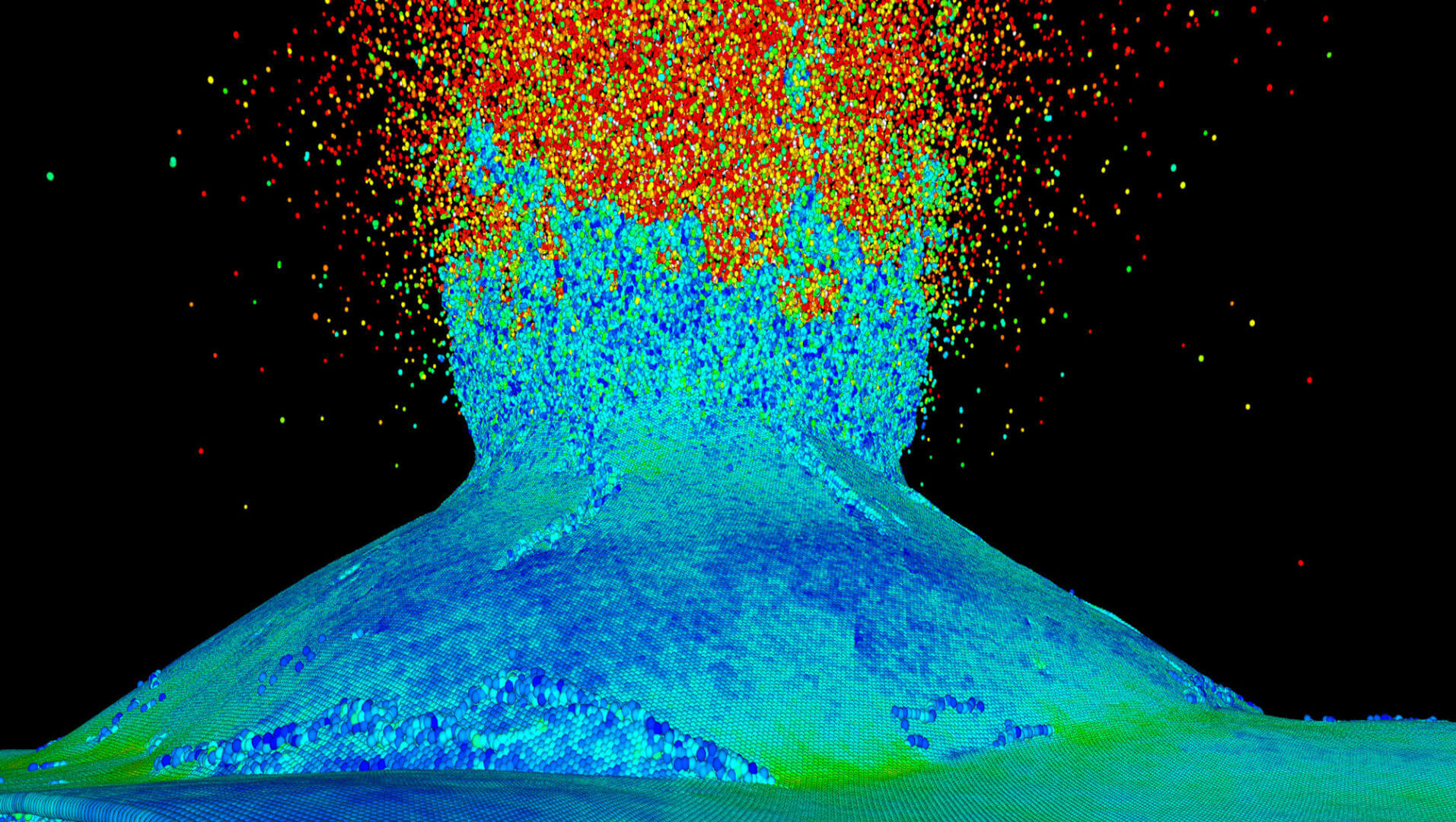
 In their supercomputer simulations, Vashishta and his fellow researchers take alumina — crystalline sapphire, a type of ceramic — and inject it with silicon carbide nanoparticles.
In their supercomputer simulations, Vashishta and his fellow researchers take alumina — crystalline sapphire, a type of ceramic — and inject it with silicon carbide nanoparticles.
They then oxygenate these nanoparticles and heat them up.
At the right temperature — around 3,000 °F — these oxidized nanoparticles then produce silica particles in a molten form, which then flows to fill the microcracks, thus “healing” the ceramic to close to its original strength, Vashishta said.
Other benefits of ceramic products include the price; they’re cheaper than metal and don’t rust or corrode, making them ideal for components in computer chips, gas turbines, fusion reactors — basically, any type of engine.
“The only thing is,” Vashishta said, gesturing to the ceramic butter plate, “the damn stuff shatters.”
Which is why self-healing is important.
Dana D. Dlott, a professor of chemistry at the University of Illinois, met Vashishta 15 years ago and was so impressed with his work that they began collaborating on several projects.
“The ultimate maturity of this self-healing technology, which requires a detailed understanding of the atomic-level processes, will lead to parts and structures that no longer break down with age, due to the inevitable formation of cracks,” Dlott said.
Vashishta gestured to the patio of the restaurant to give an idea of how micro of a world he studies.
“We’re sitting here on this large patio, and down on the ground there probably are a few ants,” Vashishta said. “But we don’t even notice them; we don’t even know they’re here.”
He compared the ants to the nanoparticles in the ceramic: miniscule parts of the whole that don’t change the material’s property in any meaningful way.
“Normally these nanoparticles in the ceramic don’t do anything. That is, until we oxygenate them and heat them to produce silica in a molten form,” Vashishta explained.
Everywhere Vashishta looked, it seemed, he saw materials.
He picked up his drink — a Gibson (gin and vermouth, garnished with an onion).
“This glass?” he said. “This is silica.”
He turned his attention back to the plate of butter — this time to put a swab of it on a roll.
“I’ve worked since age 10, and I don’t envision ever retiring,” he said. “I don’t know anything else.”